COBRE Phase I
On July 25, 2014, the Delaware Comprehensive Sickle Cell Research Center secured, under the leadership of Marie Stuart, M.D., its first 5-year funding of $10.2 million for the Program from the National Institute of General Medical Sciences (NIGMS) of the National Institutes of Health (NIH) under Award Number P20GM109021. The COBRE award is an IDeA award managed by the NIGMS. Year 1 of the 5-year grant launched on August 1, 2014 with 3 target projects, 2 pilots, and administrative as well as clinical and data management cores. Since then, the COBRE Program has awarded an additional 4 pilot awards and a gene editing core.
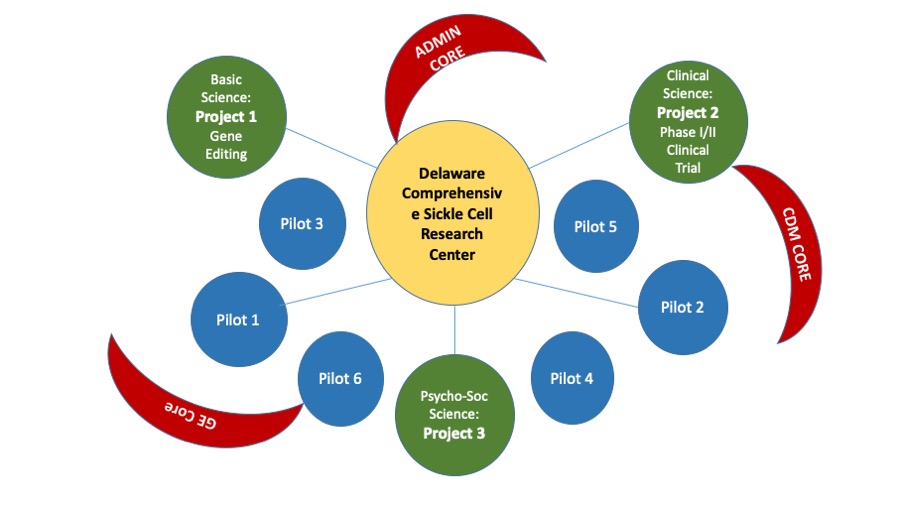
Research Projects
- Gene editing of the beta globin gene using TAL effector nucleases and single stranded oligonucleotides.
- Specialized Pro-resolving Mediators (SPMs) in Children with Sickle Cell Disease (SCD)
- Phase 1/2 clinical trial of the n-3 omega fatty acids in pediatric sickle cell disease related pain and inflammation
- Screening for psychosocial risk in pediatric sickle cell disease using the Psychosocial Assessment Tool (PAT)
Pilot projects
- Genetic risk and markers of early kidney disease in children with sickle cell disease
- In vitro growth of Stem cells on nanofiber scaffolds
- Re-expression of fetal hemoglobin by gene editing as a therapy for sickle cell disease
- The study of genomic modifiers in sickle cell disease
- Implementing Hydroxyurea Therapy among Youth with Sickle Cell Disease: A Needs Assessment
- Gene Editing in SC Disease Mutation CD34+ Cells-CRISPR/Case9 Ribonucleoprotein
Cores and Research Resources
- Administrative Core
- Clinical and Data Management Core
- Clinical Research Services
- Gene Editing Core Facility
- Access to the Gene Editing Core, Center for Translational Cancer Research, the Helen F Graham Cancer Center, Christiana Care Health System, DE
- Access to Bioinformatics and Cell Science Cores of companion COBRE at Alfred I duPont Hospital for Children
Our Program developed strong collaborations with Delaware State University (DSU), Christiana Care Health Services (CCHS) and IDeA Networks for Biomedical Research Excellence (INBRE).
